
Risk of bias in industry-funded oseltamivir trials: comparison of core reports versus full clinical study reports | BMJ Open

PDF) Assessment of Adverse Events in Protocols, Clinical Study Reports, and Published Papers of Trials of Orlistat: A Document Analysis
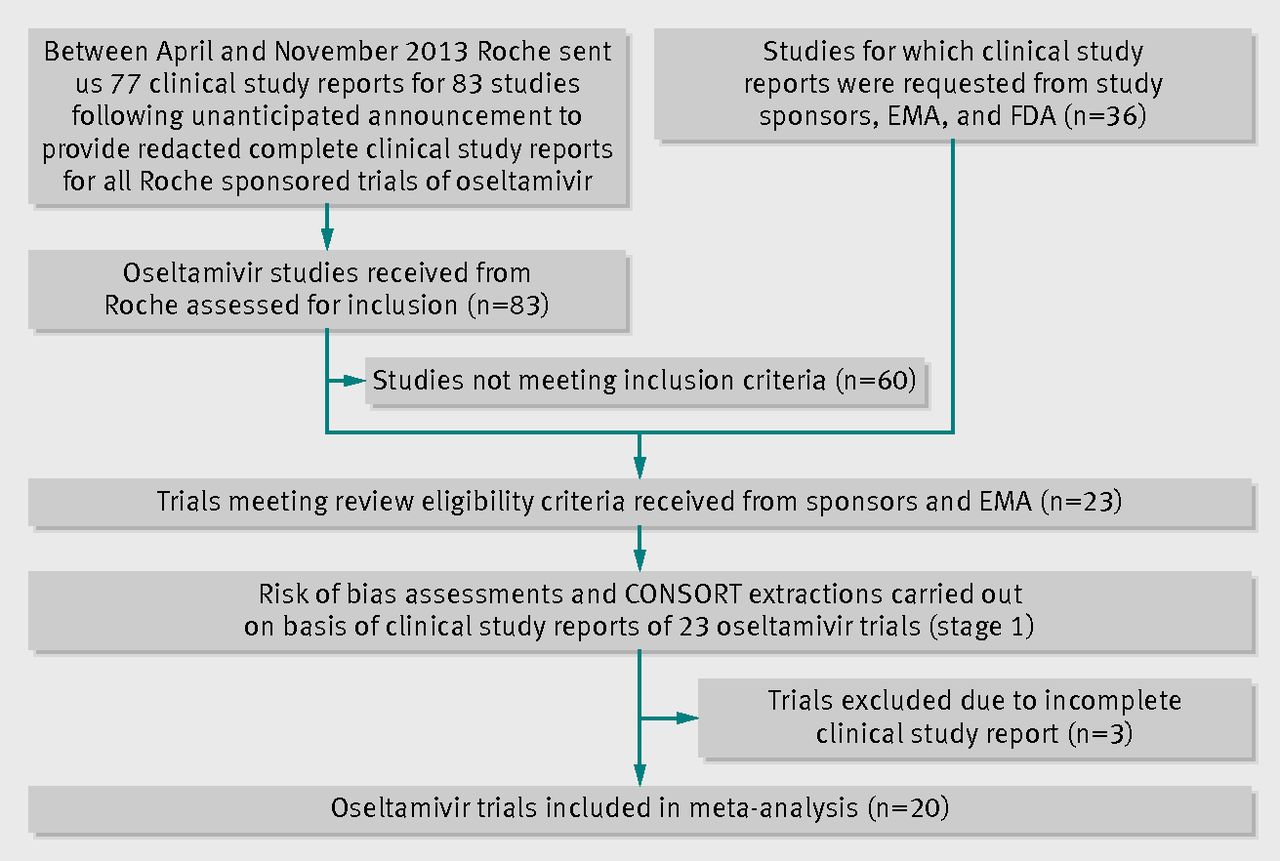
Oseltamivir for influenza in adults and children: systematic review of clinical study reports and summary of regulatory comments | The BMJ
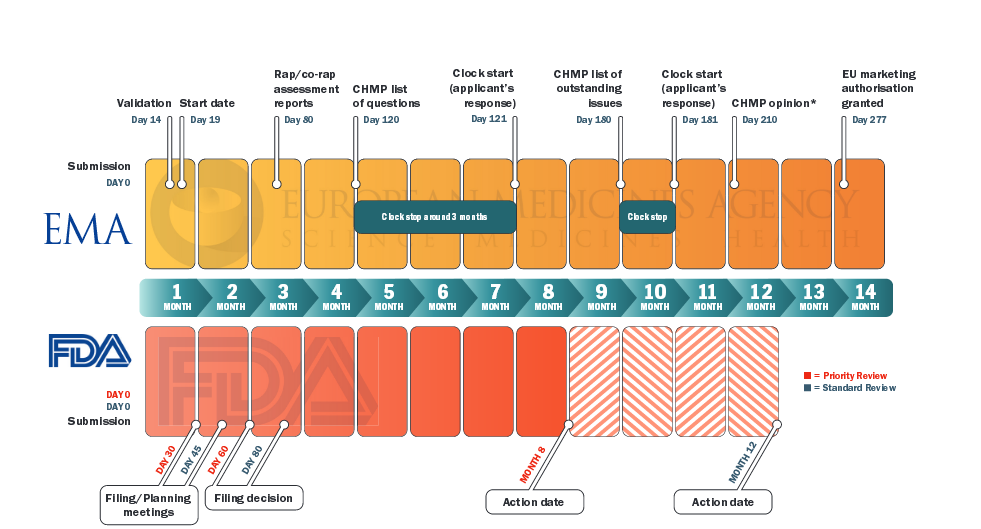
The European Medicines Agency Clinical Data Website Enables Insights Into Clinical Development Timelines And Strategy. - Document - Gale Academic OneFile

PDF) Clinical study reports of randomised controlled trials: An exploratory review of previously confidential industry reports





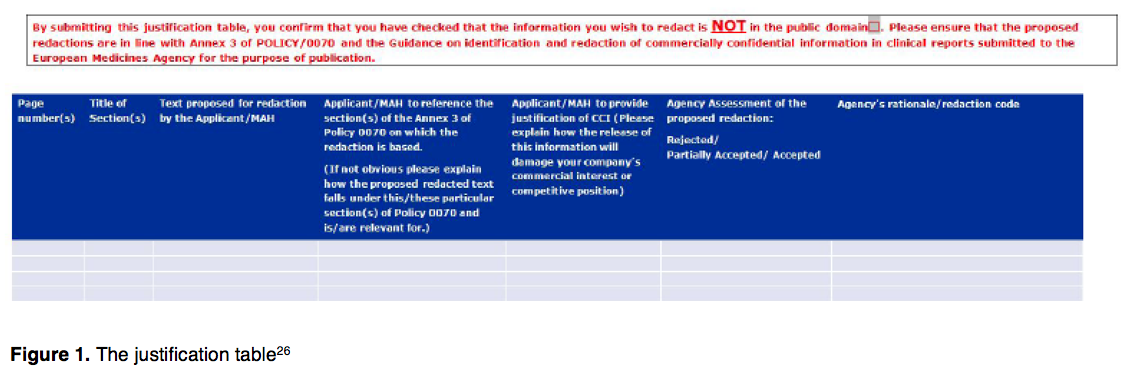




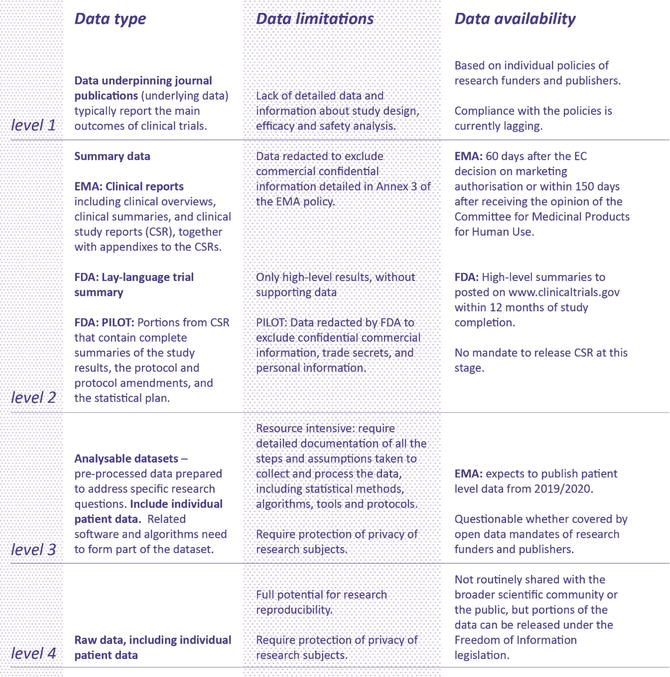


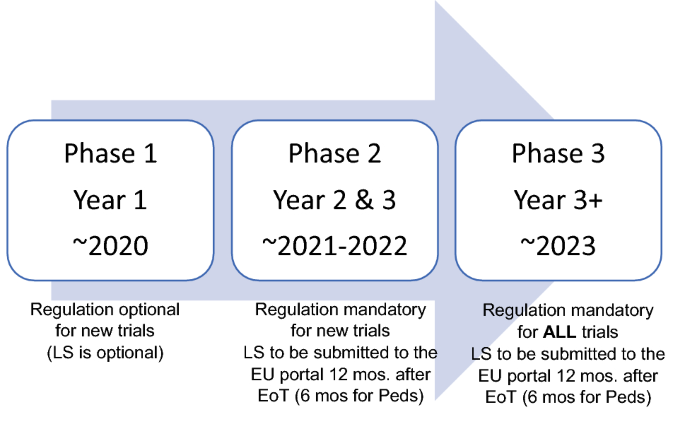
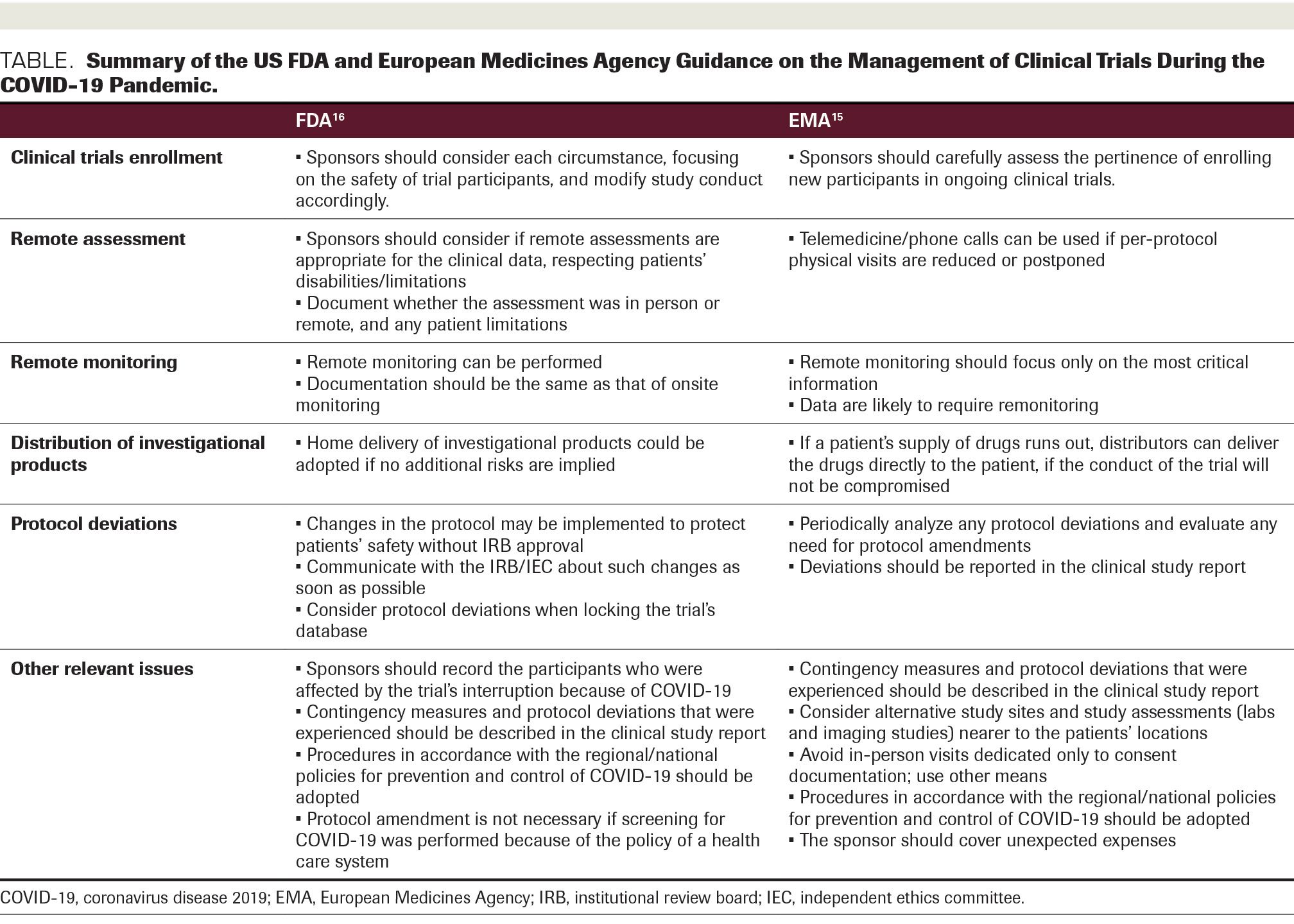


![PDF] Effective authoring of clinical study reports: A companion guide | Semantic Scholar PDF] Effective authoring of clinical study reports: A companion guide | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2f8d38c9afbd7fce7a7635cf459a7989c82a753a/5-Table1-1.png)
